|
12/25/2023 0 Comments Valence electrons of antimony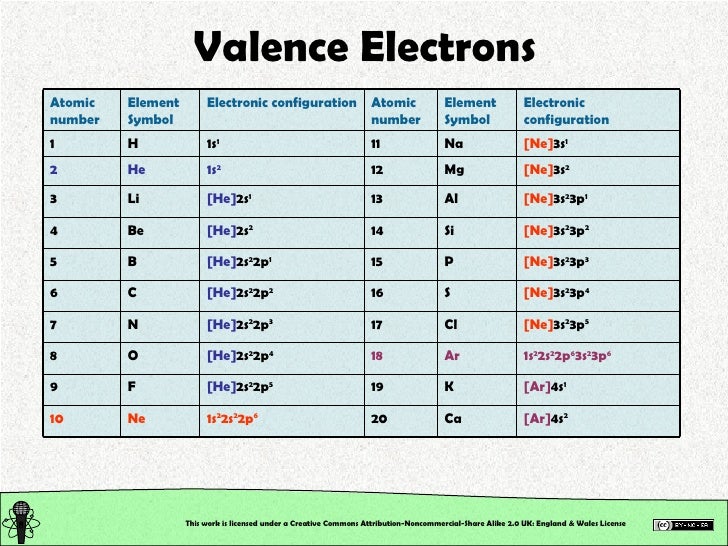
Read more about periodic trends here: brainly. 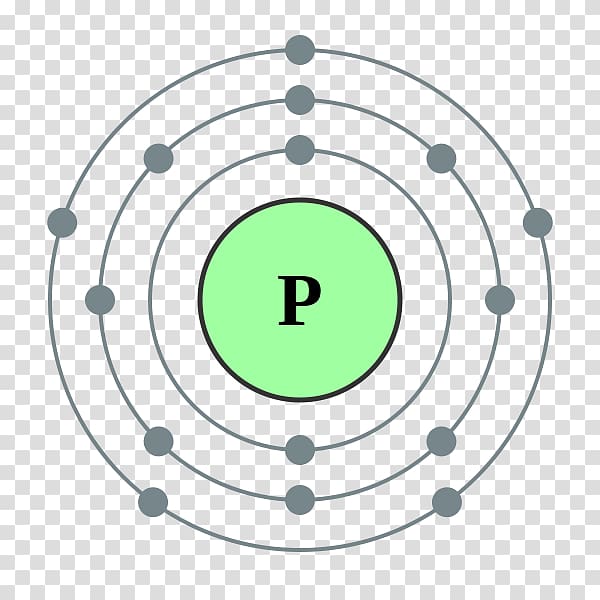
Since Antimony belongs to group 15 and period 5, its valence electrons is 5. Ions is the term for atoms that have gained or lost electrons.Īs said earlier, looking at the periodic table can help us know the number of valence electrons of an atom. That is why it is "electrically neutral" because the atom hasn't gained or lost any electrons. In these groups and periods, elements are together because of their similarities like number of valence electrons, electron affinity, and electronegativity.Įlectrically neutral atoms are atoms that contains no net charge. Elements in the periodic table are arranged in a manner where you can find them in groups and periods. One way to know the number of valence electrons present in an atom is to look at the periodic table and periodic trends. Bond order refers to the number of bonds that can be formed between two atoms. Antimony is the element of nitrogen family hence it. The precise valency of Antimony is 5 since it contains 5 valence electrons in its outer shell. You can easily understand the number and bonding of Antimony’s valence electrons in the diagram. Valence electrons also indicate the bond order of a chemical compound. It’s further useful in depicting the chemical bonding of valence electrons. They are considered important because they are responsible for some of the element’s chemical properties like the element's electronegativity. Valence electrons are electrons that lie at the outermost shell of the nucleus' electron orbit. Skip to site menu on this page Periodic Table of Elements Element Antimony - SbĬomprehensive data on the chemical element Antimony is provided on this page including scores of properties, element names in many languages, most known nuclides of Antimony.The number of valence electrons in of an electrically neutral antimony atom is 5. Common chemical compounds are also provided for many elements. In addition technical terms are linked to their definitions and the menu contains links to related articles that are a great aid in one's studies. 
Routes of Exposure: Inhalation Ingestion Skin and/or eye contact.3141 / 157 inorganic liquid compouns, n.o.s.Vapor Pressure = C Regulatory / Health.Flammablity Class: Non-combustible solid (except as dust).Enthalpy of Atomization: 263.6 kJ/mole 25☌.Description: Hard brittle bluish-white metal.Conductivity Electrical: 0.0288 10 6/cm Ω.Coefficient of lineal thermal expansion/K -1: 8.5E -6.Valence Electron Potential (-eV): 57 Physical Properties of Antimony.Incompatibilities: Strong oxidizers, acids, halogenated acids.Electronegativity: 2.05 (Pauling) 1.82 (Allrod Rochow).Electrochemical Equivalent: 1.5142g/amp-hr.Valence Electrons: 5s 2p 3 Electron Dot Model.Number of Neutrons (most common/stable nuclide): 71.Number of Electrons (with no charge): 51.Electrons per Energy Level: 2,8,18,18,5 Shell Model.Electron Configuration: 1s 2 2s 2p 6 3s 2p 6d 10 4s 2p 6d 10 5s 2p 3.Ĭross Section (Thermal Neutron Capture) σ a/ barns : 4.91.Swedish: Antimon Atomic Structure of Antimony.Series: Metalloids ( Metals) Antimony's Name in Other Languages.Common Chemical Compounds of Antimony Overview of Antimony.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
